ARTHRO·SPIN·LIFT · MBST therapy device
ARTHRO·SPIN·LIFT stands for convincing medical technology. The MBST therapy device combines therapeutic efficacy and patient comfort with a unique design and also convinces in everyday business with its functionality, user-friendliness, aesthetics and hygienic aspects. With its wide range of indications ARTHRO·SPIN·LIFT sets new standards. The therapy system is designed for the reliable, gentle and safe treatment of a variety of degenerative diseases of the musculoskeletal system. It is also used in the treatment of bone metabolism disorders and to accelerate healing processes. This makes it an ideal supplement for postsurgical, post-inpatient and rehabilitation accompanying treatment strategies for injuries and diseases of the musculoskeletal system.

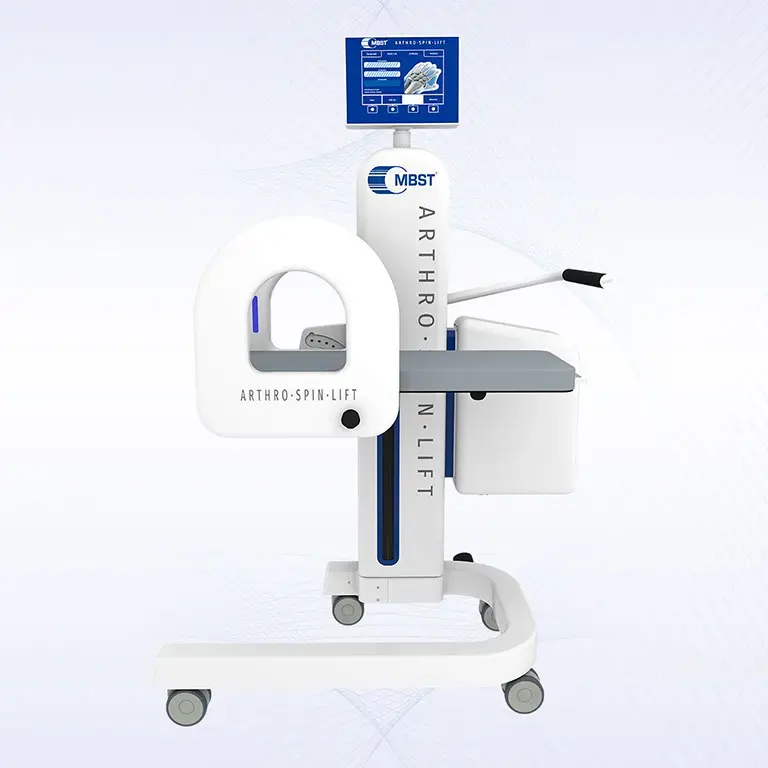
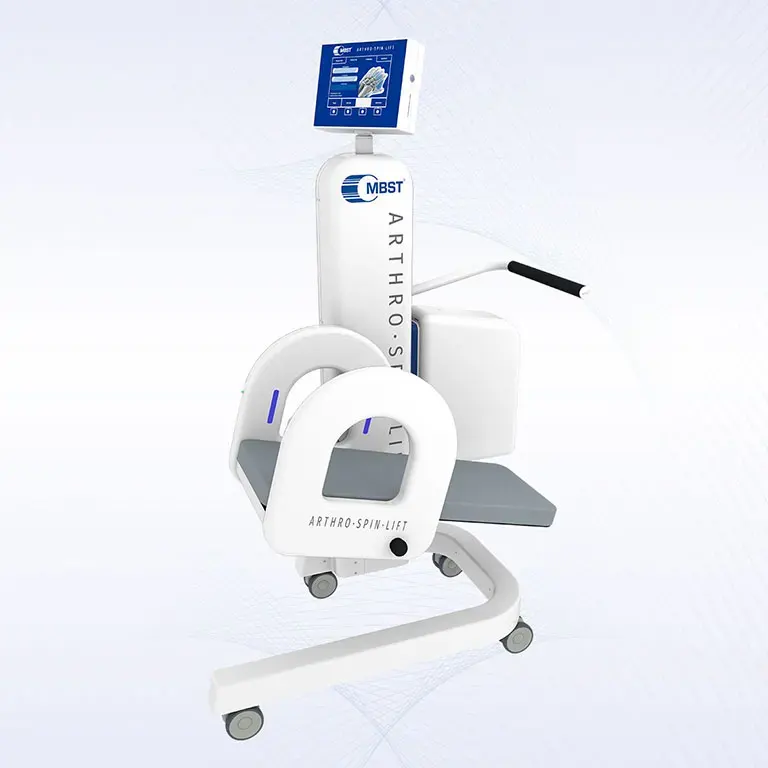
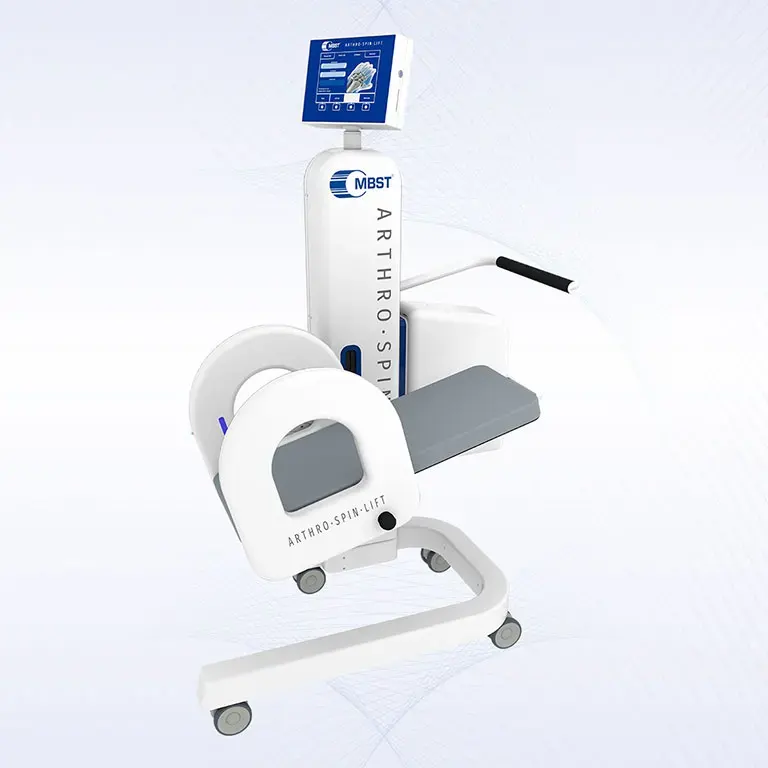
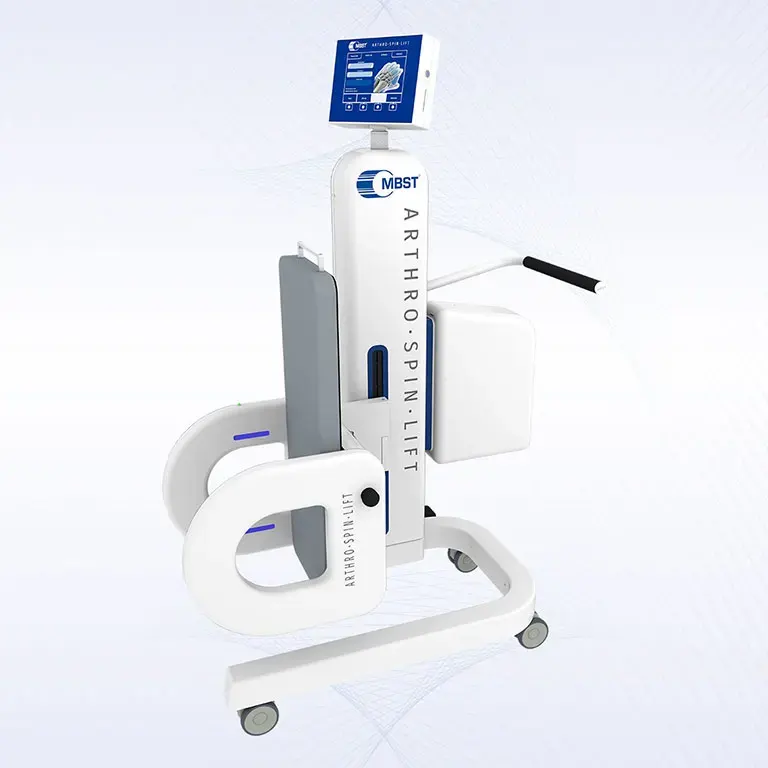
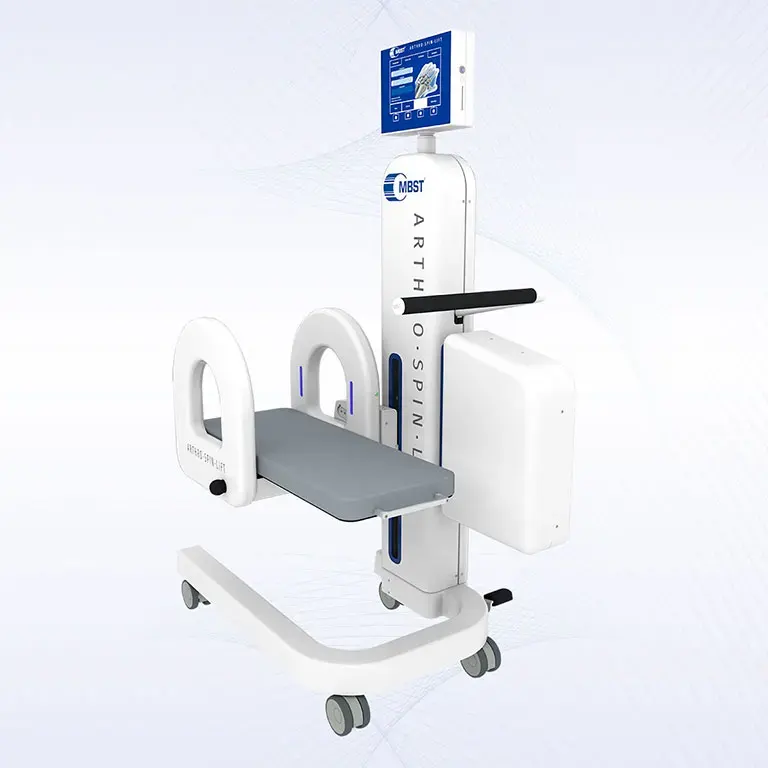

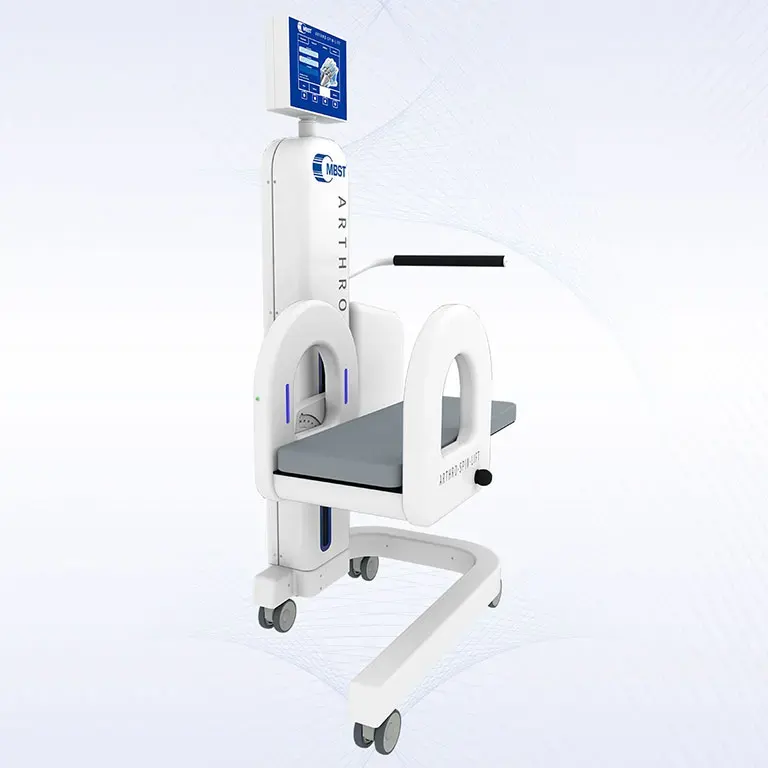
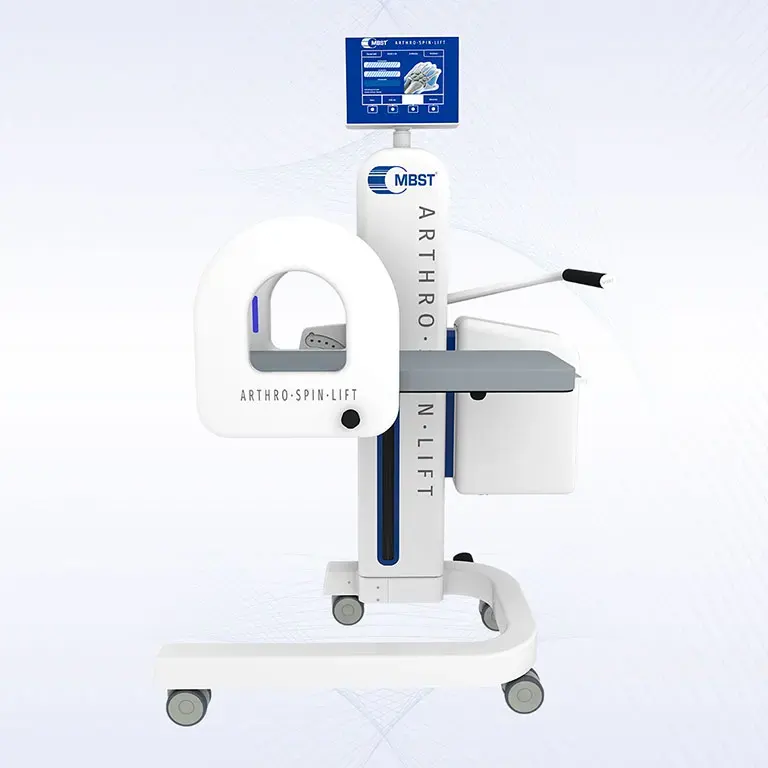
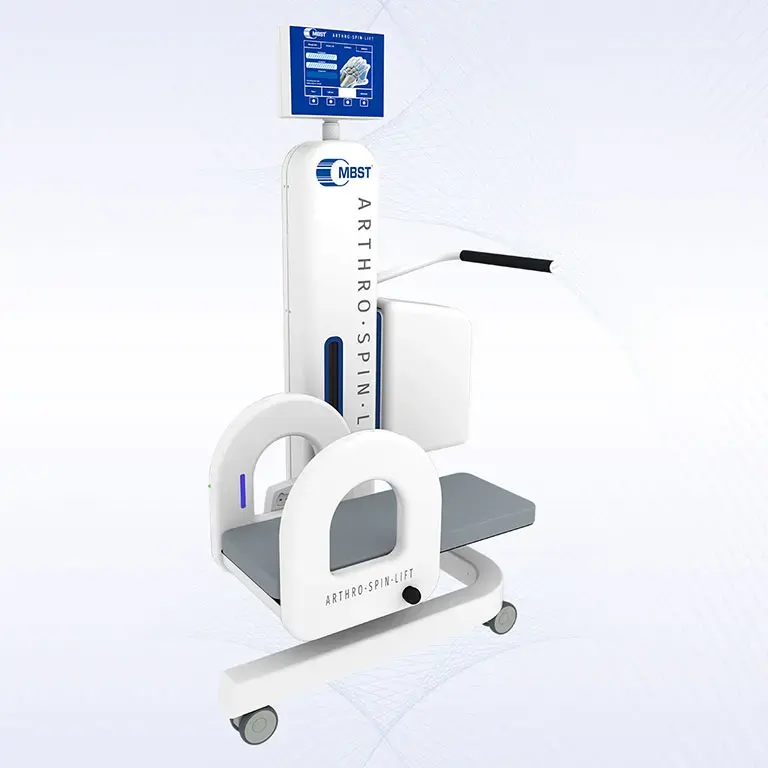
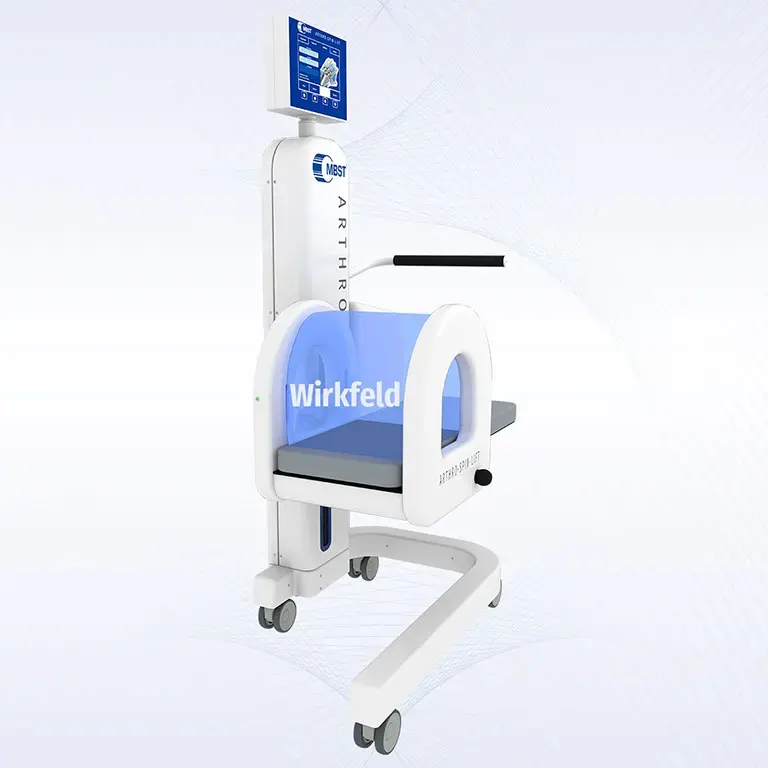




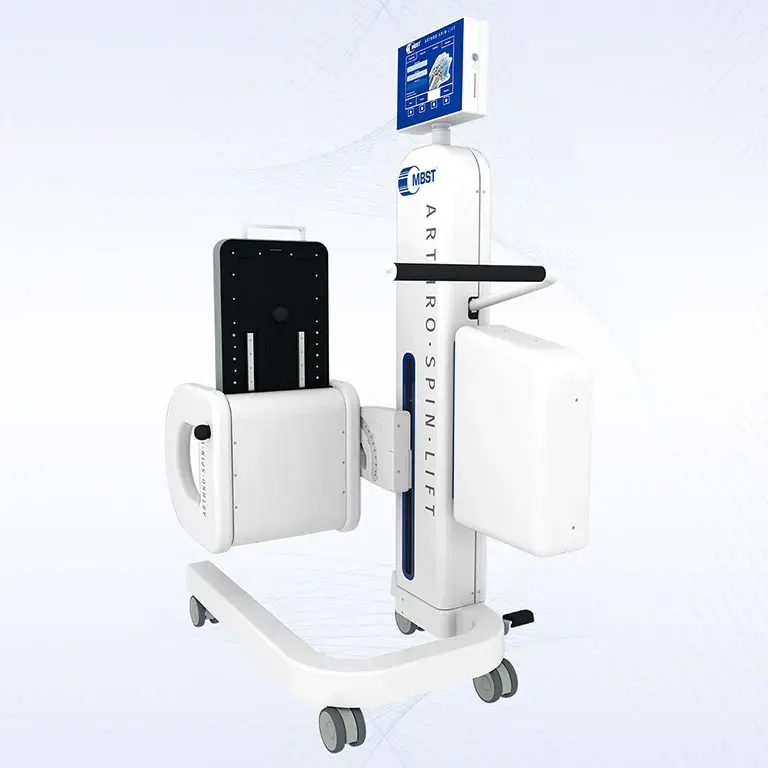















ARTHRO·SPIN·LIFT
The rollable and multi-adjustable therapy device allows the treatment of patients of almost any age and body size. It can be moved easily with small effort. The efficient treatment can be adapted optimally to each patient. ARTHRO·SPIN·LIFT takes up only a small space and thus fits even into smaller rooms. Its surfaces have no corners or sharp edges, are resistant to impacts, scratches and abrasions and easy to clean and to disinfect.
The patented medical technology of the MBST therapy device complies with current EU norms and can be used for all age groups.
The effect of magnetic resonance on the circadian cell clocks, which were identified by chronobiologists (Nobel Prize for Medicine 2017) as the cause of many diseases, is also particularly noteworthy in ARTHRO·SPIN·LIFT. New research findings show that MBST technology can actively influence the inner clockwork of cells. In this way it can practically achieve a new adjustment of the natural clock generator of our body cells. The aim is to reprogram these clocks in order to enable smooth running again. New technological developments based on these findings are filed for patent application. They show the uniqueness of MBST magnetic resonance therapy.
The most important new developments and advantages of ARTHRO·SPIN·LIFT at a glance:
| Treatment field: | approx. 33 l |
|---|---|
| Construction: | open therapy device |
Extract of the list of possible indications that have already been treated successfully with MBST magnetic resonance therapy:
Achillodynia, Ahlbaeck’s disease, baker’s cyst, bone bruise, bone marrow edema syndrome, bursitis, calcaneal spur, carpal tunnel syndrome, chondromalacia patellae, CRPS, femoroacetabular impingement, fractures with and without joint involvement, Hagelund’s deformity, hallux rigidus, hallux valgus (postsurgical), Hegemann’s disease, Hoffa-Kastert syndrome, impingement, Iselin’s disease, jumper’s knee, Kienbock’s disease, Kohler’s disease I and II, lateral epicondylitis, medial epicondylitis, meniscal lesion, microfractures, Osgood-Schlatter-disease, osteoarthritis (Bouchard nodes, gonarthrosis, Heberden nodes, osteoarthritis of the elbow, osteoarthritis of first carpometacarpal joint, polyarthrosis, posttraumatic arthrosis, rhizarthritis), osteochondrosis, osteochondritis dissecans, osteonecrosis, osteopenia, osteoporosis, Panner disease, partial ruptures of ligaments, muscles, tendons, periostitis, plantar fasciitis, Preiser’s disease, prosthesis loosening, pseudarthrosis, postsurgical (after arthroscopy, chondrocyte transplantation, joint replace ment or osteotomy), shin splints, stress fracture, tendinitis, tendinopathy, wound healing complications
All technical data contained in this publication is for product information purposes only and not legally binding. In case of further development of products, the older versions will become invalid. The current technical data will be provided by MedTec Medizintechnik GmbH upon request. Please note that technical data states only average values. All values are generated by tests according to industry standards. The results may vary due to different standard interpretations by different service providers or laboratories. All technical data applies to new products only. Deviations due to production may occur.


Information for patients about the therapy device ARTHRO·SPIN·LIFT